Analyzing the Emergence of Covid Variant KP.2 and Its Potential Impact
(Posted on Friday, May 31, 2024)
This article was originally published on Forbes on 5/31/2024.
It is clear that SARS-CoV-2, the agent of the Covid-19 epidemic, is here to stay. Like influenza, an easily transmissible respiratory virus, SARS-CoV-2 mutates to evade the immune system of those who have been previously vaccinated, infected, or both. The question remains: how dangerous is each new version of SARS-CoV-2? Again, like the Flu, if you have been previously vaccinated, infected, or both, new variants are not so dangerous, but if you have not, emerging versions of SARS-CoV-2 could be life-threatening for those vulnerable.
As Summer approaches, another series of new strains is on the horizon. While these are derived from the original Omicron and share many similarities, they are sufficiently different to spark a new wave of Covid-19 cases.
One new variant, KP.2, could lead the surge during the upcoming summer. This recently identified variant is mainly spreading in the United States, Canada, and the United Kingdom, with rising levels in Singapore, New Zealand, and Australia. As of now, there have been 1,816 reported cases of KP.2 in the GISAID SARS-CoV-2 database, indicating that potentially thousands, if not tens of thousands, of individuals, have already been infected with this variant, as sequencing efforts have been significantly limited in recent years.
KP.2 belongs to a group recently characterized as the FLiRT variants. This name is derived from the technical designations of two critical mutations in their spike proteins: the F456L mutation and the R346T mutation.
The FLiRT grouping is a subset of variants on the Omicron tree, which has primarily fueled SARS-CoV-2 infections over the previous two years. KP.2 is structured similarly to previous major Omicron variants, including BA.2.86, JN.1, and XBB.1.5.
The variability of Omicron is extensive. As with Influenza for many years, the past is prologue with the SARS-CoV-2 virus. Over time, new variants emerge containing unique mutations, not only in the spike protein, but throughout the whole of the virus genome. Below is a diagram demonstrating the web of Omicron variants that have emerged over the past several months. KP.2 and its FLiRT variant grouping are found in the top right corner, shaded in light green.
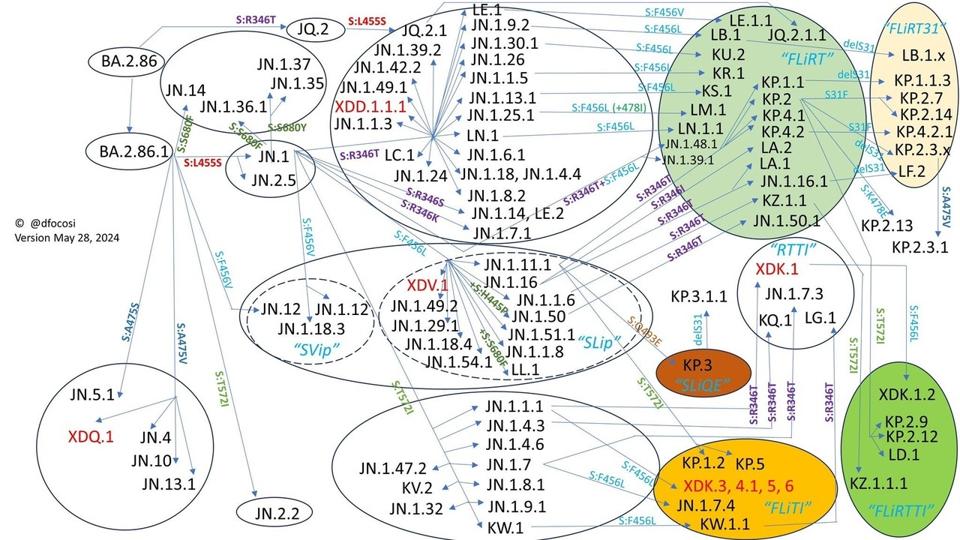
FIGURE 1: Diagram depicting the evolutionary nature of SARS-CoV-2, as demonstrated by the proliferation of Omicron variants. KP.2 and family are found in the top right corner.
As JN.1 is the most recent dominant variant, we will compare it to KP.2 throughout this article to demonstrate how KP.2 may differentiate itself from previous variants and indicate why it may ignite a new rise in cases this summer.
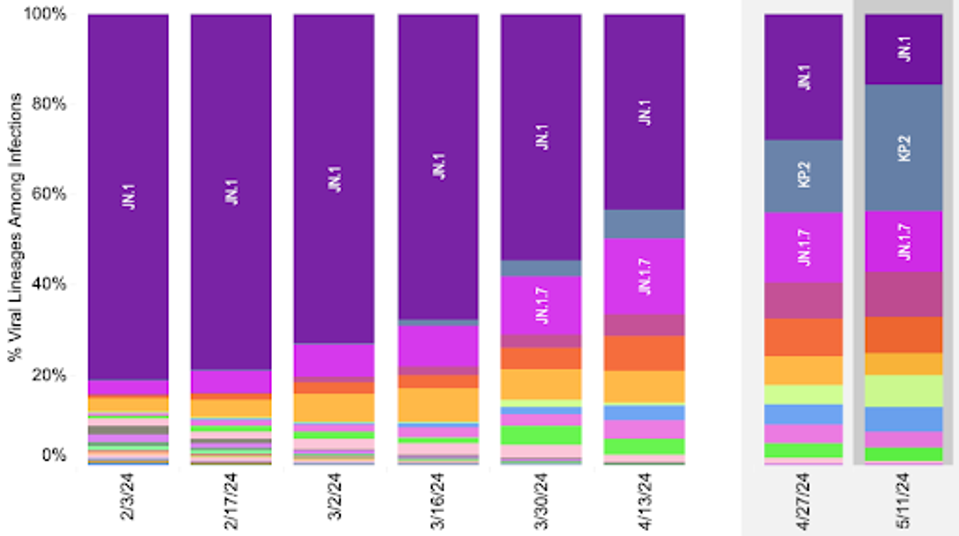
FIGURE 2: Gradual increase of KP.2 replacing JN.1 as the dominant variant in the United States.
Let’s start with the spike protein. SARS-CoV-2 needs to attach to human cells using the ACE2 receptor, merge its membrane with the host cell membrane, and ultimately gain access to start the infection and reproduction within human cells. Importantly, this protein is the main focus for vaccines, antibodies, and antiviral treatments against Covid-19.
As with JN.1, many spike protein mutations in KP.2 are also present in earlier variants of concern, including E484K and N501Y. These variants, known as the Alpha and Beta variants of SARS-CoV-2, were first detected in early 2021.
In fact, there are only three spike protein differences between KP.2 and JN.1. Two of these are the namesake of KP.2’s FLiRT grouping.
A single mutational variation exists in the N-terminal region, which is involved in virus entry after infection. In KP.2, R346T is present, which could enhance the efficiency of viral entry and enable antibody evasion by introducing N-glycosylation sites.
In the receptor-binding domain, just one change from JN.1 could significantly impact F456L, which may enhance ACE2 binding affinity or decrease antibody binding efficiency.
Finally, V1104L has been added to the S2 subunit of the spike protein, which may improve the efficiency of the fusion of the viral envelope with the host cell membrane, allowing the viral genome to enter the cell.
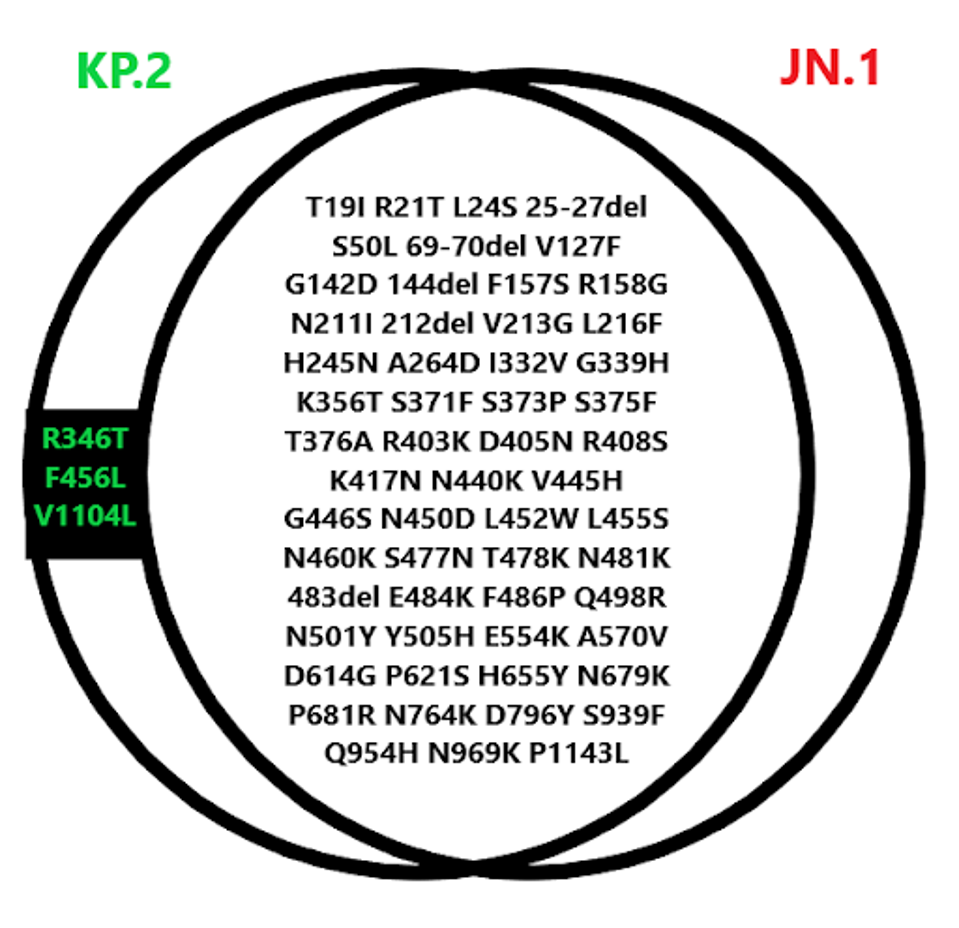
FIGURE 3: Spike protein mutational profile of KP.2 compared to JN.1.
I’d like to highlight mutations occurring outside of the spike region, which could significantly impact the virus’s ability to cause disease and spread. In various parts of the genetic material, there is a wide range of mutations in the Orf1ab replication-transcription complex (NSP1-16), some in the structural proteins (E, M, and N), and a few in the accessory proteins (Orf3a-8). We are drawing attention to these mutations because changes in specific proteins, especially the N protein, can considerably affect virus replication.
Below is the entire catalog of mutations found throughout the virus.
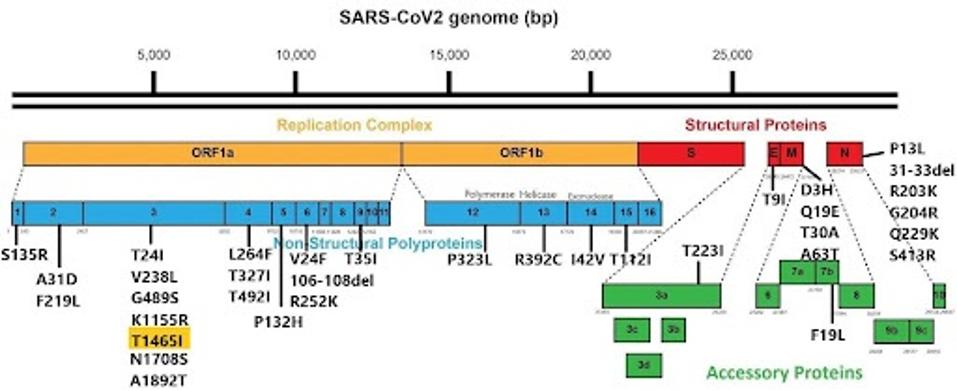
FIGURE 4: KP.2 nonspike mutation.
There is only one nonspike mutational difference from JN.1 in KP.2. It lies in the Orf1a collection, which contains 11 proteins involved in a number of replication processes. This mutation is NSP3 T1465I.
Non-structural protein three is one of the most complicated proteins in the SARS-CoV-2 virus. Among its many functions are proteolytic activity, formation of replication-transcription complexes, antagonizing host immune response, regulating viral translation, and inducing cell death. T1465I could improve any of these functions, though it is difficult to pinpoint which or to what extent.
I have noted the extensively mutated N protein. Mutations at R203K and G204R have been present in most virus variants during the pandemic and are likely to enhance the rate of viral replication. Although these mutations are identical to those in JN.1, the N protein continues to play a crucial role in the virus’s pathogenicity.
The changes in the mutations, both within and outside of the spike protein, have multiple reasons: The first reason is to adjust for increased infectivity; the second is to evade neutralizing antibodies; the third is to adapt for more effective post-infection development, which includes replication supported by mutations in the N protein; and the fourth is to avoid recognition by T cells.
Furthermore, it is worth mentioning that this virus, along with most sequenced variants throughout the Covid-19 epidemic, shares three mutations with the first departing variant from the original Wuhan virus. These are D614G in the spike, P323L in NSP12, and the C241U synonymous mutation in the 5’ end of the virus.
In fact, there are numerous synonymous mutations scattered throughout KP.2, but gathering data on these mutations is much more complex than collecting data on amino acid mutations.
The synonymous mutations do not change the virus’s amino acid sequence, but they do influence the tertiary structure of its RNA. Research indicates this can contribute to the virus’s adaptation to the human host environment. These mutations also impact the abundance of viral proteins and the immune responses in the infected individual.
It remains to be seen whether KP.2 will cause a new wave of Covid cases akin to Alpha or Omicron in years past. Further, current data cannot confirm whether this variant is more pathogenic or viral than JN.1, particularly for those previously infected or vaccinated. For those who have not been infected or vaccinated, infection with KP.2 could be very serious, and they should exercise caution.
It is crucial to be aware of these threats before they become widespread, not after. As we enter the summer months, another wave of cases could likely occur, and KP.2 is an avenue for that.