Ipilimumab, a Pioneer Breakthrough in Cancer Treatment
(Posted on Monday, May 20, 2024)
In 2011, the first in a new generation of anticancer drugs called checkpoint inhibitors was approved. The drug, known as ipilimumab or Yervoy, stood apart from standard cancer treatments. It worked by thwarting a cancer cell’s ability to fend off immune cell attacks. A new era of cancer care emerged as several other checkpoint inhibitors followed ipilimumab’s official release. Although initially designed to treat advanced melanoma, doctors today may offer ipilimumab and other checkpoint inhibitors to treat several types of cancers.
This article describes how ipilimumab works, what cancers it treats, and what developments may arise in the future.
How Ipilimumab Works
Ipilimumab (pronounced ih-pih-lih-moo-mab) is an anticancer drug delivered to the bloodstream via an intravenous line (IV). The infusion is filled with antibodies that block a particular type of protein found on the surface of immune T cells called immune checkpoints. This blocking interaction encourages white blood T cells to rally against tumors. Understanding how immune checkpoints typically work is integral to recognizing this drug’s impact.
Immune checkpoint proteins are proteins naturally found on immune T cells. These proteins are often described as an immune “braking system.” Just as brakes slow a rushing car, immune checkpoints dampen T cell activity. They safeguard against overactivation, where excessively stimulated T cells risk harming healthy tissues.
If checkpoint proteins are a natural part of the immune system, what need is there for checkpoint inhibitors?
The issue is that cancer cells co-opt this mechanism for their gain. They bind checkpoint proteins to shut off T cells that otherwise would typically retaliate against the tumor. This is one of many tactics cancer cells employ to proliferate rampantly.
Here is where checkpoint inhibitors work their magic. Ipilimumab binds to a specific checkpoint protein, blocking its access. This prevents cancer cells and other immune cells from deactivating the T cell. Liberated, the T cell is free to activate, expand and counter the tumor. Figure 1 illustrates this drug’s mechanism.
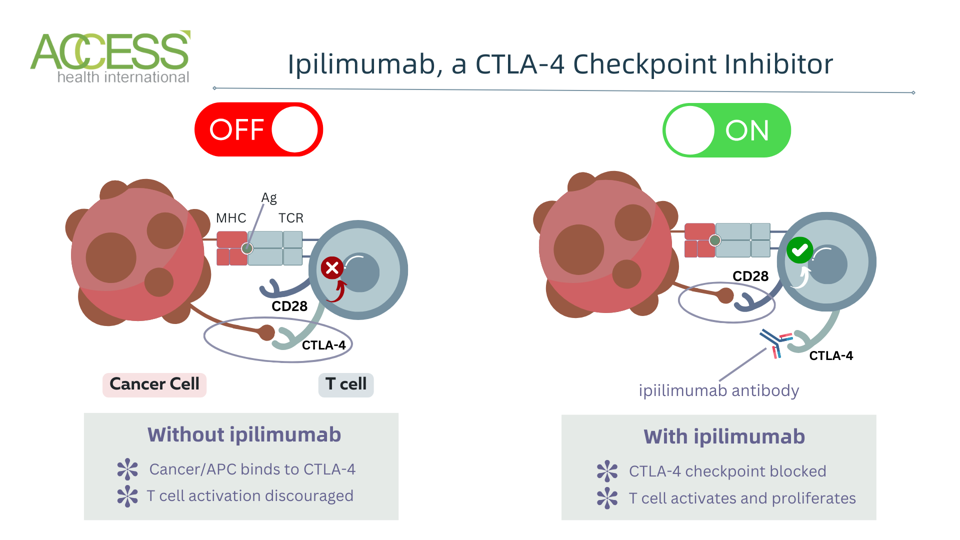
Figure 1: Ipilimumab checkpoint inhibitor mechanism. The ipilimumab antibody blocks cytotoxic T lymphocyte-associated protein 4 (CTLA-4) checkpoints on T cells. This prevents cancer and antigen-presenting cells (APC) from binding and sending an inhibitory signal. With the checkpoint blocked, the CD28 receptor can bind and send a costimulatory signal; the T cell can activate, proliferate, and enhance antitumor responses. Abbreviations: MHC, major histocompatibility complex; TCR, T cell receptor
ACCESS HEALTH INTERNATIONAL
Why Ipilimumab is Unique
Ipilimumab operates uniquely from other checkpoint inhibitors. Among all the checkpoint inhibitors on the market, ipilimumab is the only federally approved inhibitor to target a specific checkpoint protein called CTLA-4, or cytotoxic T lymphocyte-associated protein 4. As this protein operates differently from other immune checkpoints, ipilimumab evokes a different antitumor response than other checkpoint inhibitors.
The only other checkpoint inhibitor that targets this protein is tremelimumab (Imjudo) by AstraZeneca; however, unlike ipilimumab, it has a very conditional use. Tremelimumab must be given alongside a different checkpoint inhibitor to treat people with surgery-resistant liver cancer.
When is Ipilimumab Used?
Ipilimumab can treat several solid tumors. However, when and what it is administered with depends on the context.
While ipilimumab can be given as a monotherapy, most times it is combined with other checkpoint inhibitors or traditional cancer treatments. The drug is considered a first-line treatment for many difficult-to-treat cancers—cancers that cannot be removed entirely through surgery alone or have spread from where they initially formed.
The immunotherapy initially received federal approval to treat aggressive melanoma. Today, it is considered a first option for those with tumors that resist surgery or have spread widely. If the tumor is operable, clinicians can order ipilimumab after surgery to prevent cancer recurrence. People with melanoma typically receive an infusion once every three weeks, for a total of four times. Post-operation, patients may require less frequent infusions for up to three after the procedure.
For all other cases, ipilimumab is usually given alongside nivolumab (opdivo), another checkpoint inhibitor by Bristol-Meyer Squibb that targets a distinct checkpoint protein called PD-1. The synergistic combination elicits improved outcomes compared to ipilimumab alone. Patients begin with ipilimumab and nivolumab infusions for a couple of weeks before switching to nivolumab alone. Cancers treated with this combination include:
- Colorectal cancer
- Esophageal squamous cell carcinoma
- Renal cell carcinoma (Kidney cancer)
- Hepatocellular carcinoma (Liver cancer)
- Malignant pleural mesothelioma (Lung cancer)
- Non-small cell lung cancer
Possible Adverse Effects
All medicines can cause adverse effects. For checkpoint inhibitors, most unwanted reactions result from stimulating the immune system. The most common symptoms caused by ipilimumab include fatigue, diarrhea, fever, skin rash, colon inflammation, decreased appetite, dizziness, and constipation. Additionally, combination treatment with nivolumab may spark headaches, vomiting, muscular and abdominal pain, increased liver enzyme levels and lowered thyroid hormone levels.
These immune-related adverse events can turn severe or life-threatening if not managed promptly. For this reason, bloodwork is required before every session, and clinicians observe patients during and after each infusion.
Future Directions
Ongoing clinical trials continue to explore new applications for ipilimumab, often in conjunction with nivolumab or other cancer treatments. There are efforts to optimize dosing and scheduling regimes, minimize adverse effects, and improve efficacy. While ipilimumab has shown limited efficacy as a single agent for certain cancers like pancreatic cancer, it demonstrates more promising antitumor activity in combination regimens for various solid tumors. Research evaluating ipilimumab plus nivolumab, chemotherapy or radiation includes cancers such as small lung cell cancer, prostate cancer and bladder cancer.
Takeaways
Ipilimumab represents a revolutionary breakthrough in cancer treatment. This trailblazing checkpoint inhibitor was the first of its kind, and even today remains singular among its competition. This medicine proved that immunotherapies of this nature can unleash the body’s anticancer forces, providing an alternative means to counter solid tumors. While the therapy demonstrates limited efficacy as a single agent, it can be coupled with other checkpoint inhibitors or cancer modalities to boost antitumor responses. Continued research will explore the boundaries of this drug’s abilities and expand its therapeutic reach.
This article joins a growing series on mono cancer treatments, including novel immunotherapies such as CAR T therapy and checkpoint inhibitors. Find more at www.williamhaseltine.com.
Read the article online on Forbes.

