New Studies Highlight Promising Candidates For Second-Generation Covid-19 Vaccines
(Posted on Tuesday, July 20, 2021)
GLASGOW, SCOTLAND – DECEMBER 08: Grace Thomson receives the coronavirus vaccine from Paula McMahon at the Louisa Jordan Hospital as the roll out begins on December 8, 2020 in Glasgow, Scotland. A batch of 65,000 doses of the Pfizer BioNTech vaccine
GETTY IMAGES
The first generation of Covid-19 vaccines has performed more spectacularly than we ever could have hoped. But as more variants of SARS-CoV-2 emerge that spread faster and hit harder than their predecessors, it has become clear that the mRNA vaccines created by Pfizer and Moderna and adenovirus vaccines of AstraZeneca might not be enough to stem the incoming tide. While distribution of these vaccines should continue full speed ahead, the time is ripe for us to take a step forward and ask, what’s next?
Second-generation Covid-19 vaccines will hopefully build upon the accomplishments of their forebears in a number of ways. Of first and foremost concern is their ability to protect against any current and future variants, no matter how infectious or virulent. They should also continue to reduce viral load and elicit high titers of neutralizing antibodies. And perhaps more importantly, if they’re to play a role in redressing vaccine shortages across the globe, second-generation vaccines should be cheap and convenient to produce and administer en masse.
Two subunit vaccines, only recently brought to our attention by a pair of just published research papers, are contenders that have shown particular promise in animal models. Unlike vaccines that use live virus to initiate immunization, subunit vaccines—such as those available for hepatitis B and the human papillomavirus (HPV)—are loaded with specific bits of the virus particle, which being only fragments usually require an adjuvant, or booster shot, to amplify the subsequent immune reaction. While other vaccine types can cause complications or adverse effects in people with chronic immunodeficiencies, nearly everyone can withstand the immune response a subunit vaccine triggers—a general level of tolerance suited to epidemics of global proportions.
Both subunit candidates are created using the receptor binding domain, a region of the spike protein of SARS-CoV-2 that facilitates attachment to human cells. The scientists who developed the first coupled a yeast-based vaccine manufacturing platform with the adjuvant 3M-052, a synthetic molecule that activates the immune signaling proteins called toll-like receptors 7 and 8. The results, available to read in Science Immunology magazine, show that in rhesus macaques, the RBD+3M-052-alum vaccine succeeded at stimulating neutralizing antibodies as early as four weeks following a first shot; reducing the number of viral particles that replicate in the respiratory tract; and lessening the severity of any lung disease. The yeast-expression technology also happens to be easily scalable—ideal, in other words, for large-scale production.
For the second subunit vaccine, its corresponding paper published in the journal ACS Infectious Diseases last week, a team of researchers used glycan engineering to create a multimeric derivative of the SARS-CoV-2 receptor-binding domain, then fused it to a trimerization motif taken from a human cartilage matrix protein (hCMP). Administered to mice and guinea pigs in tandem with a squalene-in-water emulsion adjuvant, the hCMP-mRBD vaccine proved highly immunogenic, generating titers of neutralizing antibodies that surpassed those of natural infection (more on this shortly). Some of the best Covid-19 vaccines we already have on hand have one major limitation: they perish if not stored at ultra-low temperatures. But this so-called “warm vaccine” is designed to have a high tolerance for heat. According to the study, it remains stable for up to a month at 37 degrees Celsius (98.6 degrees Fahrenheit).
From that point of comparison we arrive at another—how the immune responses triggered by the subunit vaccines compare to those of the preceding generation, not just against the original SARS-CoV-2 strain but the new variants that have emerged since. Making a one-to-one comparison is difficult due to differences in experimental design. But examining the fluctuations in antibody titer documented from study to study, we can gather that subunit vaccines might perform as well as our current mRNA vaccines.
The antibody titers elicited in monkeys by the first subunit vaccine, RBD+3M-052-alum, reached levels comparable to mRNA vaccines against the Alpha (B.1.1.7) and Beta (B.1.351) variants. Also like mRNA vaccines, the antibody titers generated by the subunit vaccines were more robust than those produced through natural infection. In mice and guinea pigs the second, heat-tolerant subunit vaccine, hCMP-mRBD, generated neutralizing antibody counts higher than those of convalescent sera, both against the original strain and the Beta variant.
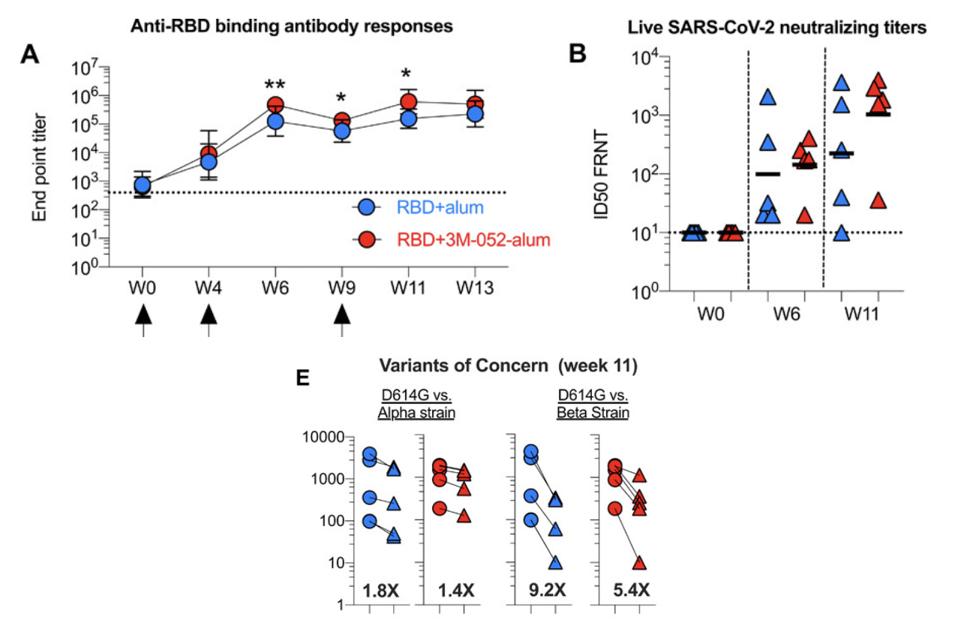
Red dots indicate antibody titers generated by RBD+3M-052-alum subunit vaccine
“A YEAST EXPRESSED RBD-BASED SARS-COV-2 VACCINE FORMULATED WITH 3M-052-ALUM ADJUVANT PROMOTES PROTECTIVE EFFICACY IN NON-HUMAN PRIMATES” HTTPS://IMMUNOLOGY.SCIENCEMAG.ORG/CONTENT/6/61/EABH3634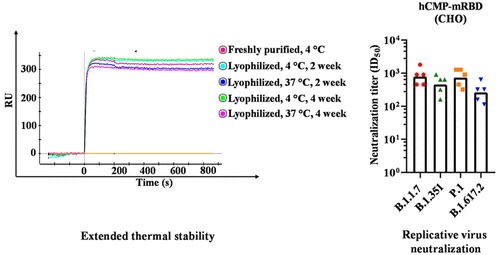
On the right: antibody titers generated by hCMP-mRBD vaccine in guinea pigs
“IMMUNOGENICITY AND PROTECTIVE EFFICACY OF A HIGHLY THERMOTOLERANT, TRIMERIC SARS-COV-2 RECEPTOR BINDING DOMAIN DERIVATIVE” HTTPS://PUBS.ACS.ORG/DOI/10.1021/ACSINFECDIS.1C00276Pfizer and Moderna’s mRNA vaccines represent the cream of the current crop—and many low- to middle-income countries have only restricted access to both. Their reserves are stocked with Covid-19 vaccines that still possess protective capabilities, but pack a more modest punch, especially against new SARS-CoV-2 variants. According to one study, against the Beta variant, the ability of the Pfizer-BioNTech vaccine to protect from severe illness and death remains at 97 percent. For the AstraZeneca vaccine, according to another study, that percentage is just 10.4.
The Delta variant, which now accounts for the lion’s share of new Covid-19 infections in many countries, is even more infectious and lethal than the Beta variant—yet another reason why we need more tools in our arsenal capable of generating vigorous immune protection. We can expect clinical trials for at least one candidate, the heat-tolerant hCMP-mRBD vaccine, to be held later this year. My hope is they arrive at our doorstep before a variant even more infectious than Delta gets there first.
Originally published on Forbes (July 20, 2021)

